Please provide a simple explanation.
Answer
Best
You’ve probably heard of a “heat pump”. You can read further on how this works exactly. A heat pump does exactly what the name suggests: pumping heat from one place to another. In the case of a freezer, the heat is pumped out from the inside and transferred to the outside. A heat pump for heating a house does the opposite, for example the heat from groundwater is pumped into the house. If you reverse this system, you can also cool your house with the same heat pump and pump the heat from the inside to the outside.
The question that now arises is: how do you get heat from a place where it is -15°C (in the freezer) to a place where it is +20°C?
You may have noticed that when you come into contact with a liquid that evaporates easily (rubbing alcohol, ether, acetone) it can feel very cold when the temperature is originally the same as the ambient temperature. A liquid always needs heat to evaporate, that heat is extracted from your skin by these liquids. The faster a liquid evaporates, the more heat it will extract from the environment, so the colder it feels. This is also why we sweat. By evaporating sweat (which starts at a temperature of 37°C when it comes out of our pores), heat is withdrawn from our body and our body cools down. When you are sweating in the airflow, it feels even colder because the sweat can evaporate even faster. The reverse is also valid: when a substance in gas form changes to liquid form (= condensation), extra heat is released. It is for this reason that burns from steam at 100°C can be more serious than burns from water at 100°C. (of course the amount of water and/or steam plays a major role here, but I’ll leave that out for now)
Specifically, for the freezer, a gas is used that takes on both the gas phase and the liquid phase during the operation of the freezer. This gas is started to be pressurized to a higher pressure by using an electrically driven pump (= the compressor). As a result, this gas is at a higher pressure and at the same time at a higher temperature. Suppose this gas now has a temperature of +50°C, then by using a kind of radiator (the condenser) this gas can still lose its heat to the air in the environment, outside the freezer, which is heated at 20°C. state. This usually happens on the back of the freezer or refrigerator, this is the black grid. The gas that is used for this application has been chosen in such a way that it changes to liquid form at that higher pressure and a meanwhile decreased temperature of, for example, 30°C. So a state change occurs. The advantage of this change of state is that the substance will release even more energy and therefore even more heat is released to the outside. When we now bring this gas (which has meanwhile assumed the form of liquid) back to the inside of the freezer (in pipes of course) and we ensure that the pressure decreases, the liquid will transfer back to the gas form and thus heat from the environment because the temperature has become much lower due to evaporation, e.g. -30°C. Now heat can transfer from the inside of the freezer (-15°C) to the gas (-30°C). The gas returns to the compressor and the story begins again.
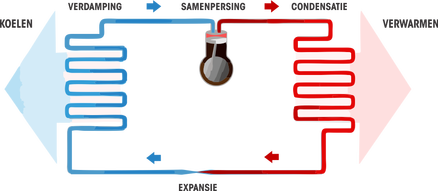
figure: http://www.ameel-technics.be/heatpumps.html
Hopefully this may clarify the operation of a freezer or refrigerator.
If you still have questions, don’t hesitate to contact me.
Kind regards,
Dirk Merckx
Answered by
ing. Dirk Merckx
Automation Pneumatics Industrial Hydraulics PLC Programming Machine Safety

Brothers De Smetstraat 1 9000 Ghent
http://www.odisee.be
.